While both Siberian Huskies and Finnish Lapphunds are generally healthy breeds, they do have the occasional health issue that comes up. In order to ensure the incidence of health problems stays low, it is important that breeders monitor their lines and test their breeding stock for known health issues. Health testing is an important part of breeding healthy dogs, but what the breeder does with that information is equally as important. A dog that passes all of it's health tests is not necessarily healthy so talk to your breeder about the general health and wellbeing of the parents and family members of your potential puppy. His or her parents may seem healthy but if they have had puppies in the past or have family members with health issues, you may be better to look for a puppy elsewhere. Do your research, ask loads of questions and above all, find a breeder you trust. While you may want a puppy as soon as possible, waiting for the right puppy from the right breeder will save you a lot of heartache in the future.
In Finnish Lapphunds, basic health testing for breeding dogs includes:
- hip and elbow scoring (to test for hip and elbow dysplasia),
- eye checks (for a myriad of eye conditions)
- a genetic test for PRCD-PRA status, however this genetic test may not be done if the parents of the dog are both clear of the defective gene.
- a genetic test for GSD-II or "Pompes" disease, though this also may not be needed if the dog is clear by parentage.
Other tests may include an echocardiogram (an ultrasound of the heart) and a genetic profile.
In Siberian Huskies, basic health testing for breeding dogs is similar and includes:
- hip and elbow scoring,
- eye checks and
- a genetic test for XL-PRA status except where the dogs are clear by parentage.
In Finnish Lapphunds, basic health testing for breeding dogs includes:
- hip and elbow scoring (to test for hip and elbow dysplasia),
- eye checks (for a myriad of eye conditions)
- a genetic test for PRCD-PRA status, however this genetic test may not be done if the parents of the dog are both clear of the defective gene.
- a genetic test for GSD-II or "Pompes" disease, though this also may not be needed if the dog is clear by parentage.
Other tests may include an echocardiogram (an ultrasound of the heart) and a genetic profile.
In Siberian Huskies, basic health testing for breeding dogs is similar and includes:
- hip and elbow scoring,
- eye checks and
- a genetic test for XL-PRA status except where the dogs are clear by parentage.
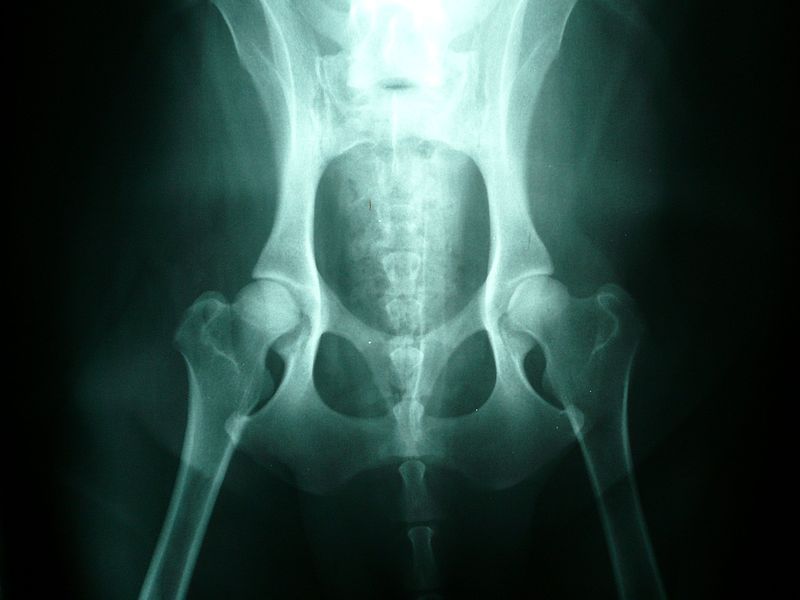
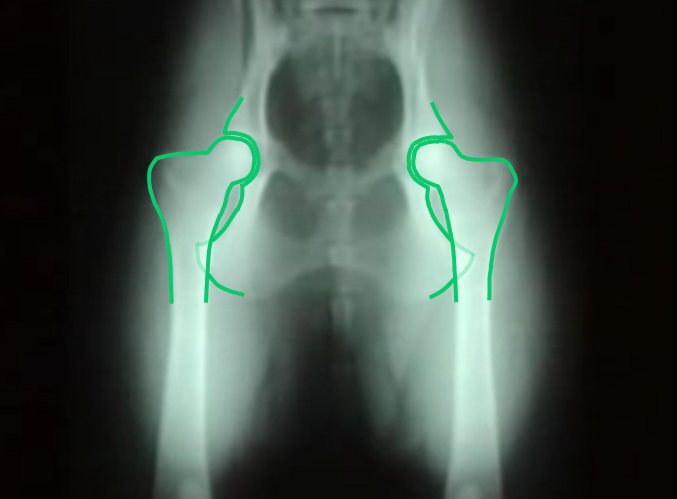
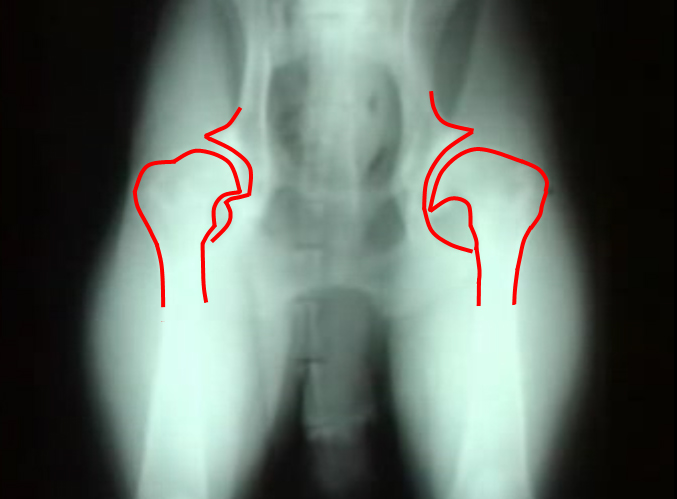
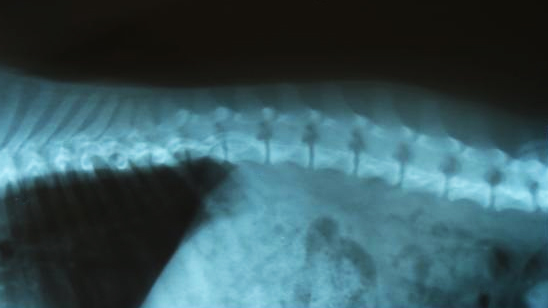
Hip Dysplasia
Hip dysplasia is an abnormal formation of the hip socket that, in its more severe form, can eventually cause crippling lameness and painful arthritis of the joints. It is a genetic (polygenic) trait that is affected by environmental factors. It can be found in many animals and in humans, but is most commonly associated with dogs, and is common in many dog breeds, particularly the larger breeds.
Normal hip anatomy In the normal anatomy of the hip joint, the root (the thigh bone) is connected to the pelvis at the hip joint. The almost spherical end of the femur head (the caput, or caput ossis femoris) fits into the acetabulum (a concave socket located in the pelvis). The bony surfaces of the femur head and of the acetabulum are covered by cartilage. While bones provide the strength necessary to support body weight, cartilage ensures a smooth fit and a wide range of motion. Normal hip function can be affected by congenital conditions such as dysplasia, discussed in this article, trauma, and by acquired diseases such as osteoarthritis and rheumatoid arthritis. Dysplastic hip anatomy In a hip suffering from dysplasias, two things are commonly abnormal. First, the caput is not deeply and tightly held by the acetabulum. Instead of being a snug fit, it is a loose fit, or a partial fit. Secondly, the caput or acetabulum are not smooth and round, but are misshapen, causing abnormal wear and tear or friction within the joint as it moves. The body reacts to this in several ways. First, the joint itself is continually repairing itself and laying down new cartilage. However, cartilage repair is a relatively slow process, the tissue being avascular. So the joint may suffer degradation due to the abnormal wear and tear, or may not support the body weight as intended. The joint becomes inflamed and a cycle of cartilage damage, inflammation and pain commences. This is a self-fueling process, in that the more the joint becomes damaged, the less able it is to resist further damage. The inflammation causes further damage. The bones of the joint may also develop osteoarthritis, visible on an X-ray as small outcrops of bone, which further degrade the joint.[2] The underlying deformity of the joint may get worse over time, or may remain static. A dog may have good X-rays and yet be in pain, or may have very poor X-rays and have no apparent pain issues. The hip condition is only one factor to determine the extent to which dysplasia is causing pain or affecting the quality of life. In mild to moderate dysplasia it is often the secondary effects of abnormal wear and tear or arthritis, rather than dysplasia itself, which is the direct causes of visible problems. |
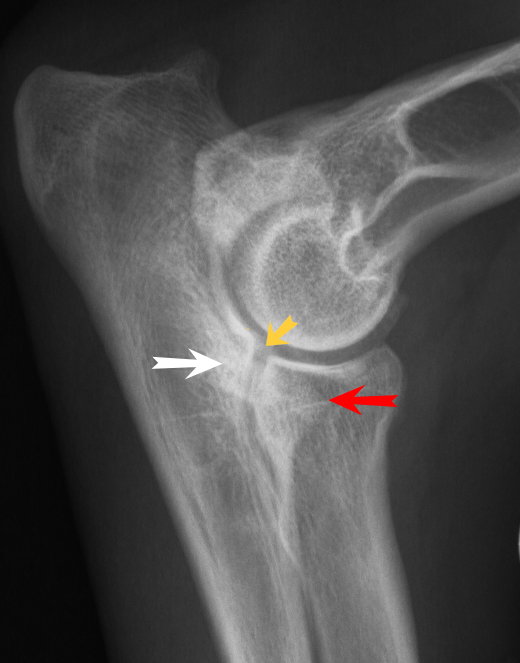
Elbow Dysplasia
This term refers to a range of conditions which involve the malformation of the elbow joint which can occur alone or in combination. These include ununited anconeal process (UAP), fragmented medial coronoid process (FCP) and osteochondrosis of the medial condyle of the humerus (OCD). Degenerative joint disease of the elbow is likely to develop from these malformations. Like with hip dysplasia, both genetic and environmental factors play a part.
Scoring for elbow dysplasia provides a number for each elbow from 0 to 3. A 0 score is provided where there is no sign of arthrosis. A score of 1 indicates minimal arthrosis, 2 indicates moderate arthrosis and 3 severe arthrosis. There is little evidence of an elbow dysplasia problem in the Finnish Lapphund breed, although the occasional case does arise. Traditionally there has not been much elbow scoring of Finnish Lapphunds, this is happening more and more, particularly in Australia, the USA and in Finland. In the majority of cases the score is 0/0. However some 0/1, 1/0 or 1/1 scores are seen, and only very rarely a worse result. |
Progressive Retinal Atrophy (PRA)
As with most cells in the body, the eyes’ vision receptors are in a continual process of renewal and replacement. In dogs affected by PRA, this process slows down and eventually, stops. In the first stages of the disease, the night vision receptors begin to fail, followed by the day vision receptors, leading to complete blindness in both eyes.
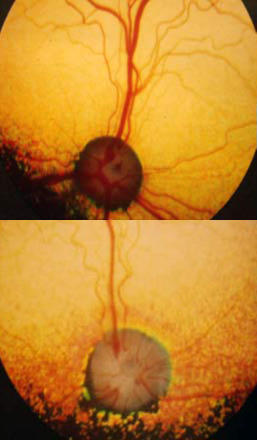
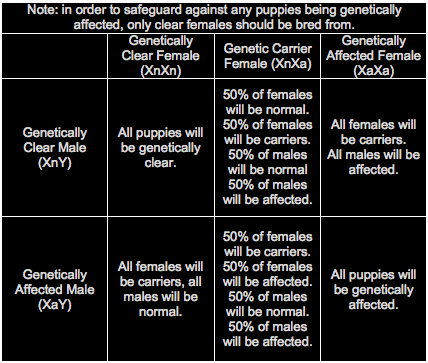
The pupils of the eye become increasingly dilated in an attempt to let in more and more light. As this happens, the eyes begin to take on a characteristic “shine”. Dogs then develop tunnel vision, losing their peripheral vision, which then narrows until they lose all sight. There are several different kinds of Progressive Retinal Atrophy, more commonly known as PRA, affecting different breeds, with different rates of degeneration. X-Linked type 1 or XL-PRA1 can be found in Siberian Huskies and Progressive Rod Cone Degeneration or PRCD-PRA found in Finnish Lapphunds. XL-PRA1 (X-linked Progressive Retinal Atrophy Type 1) – XL-PRA1 usually does not affect dogs until they are 3-5 years old, when the first noticeable clinical signs begin to manifest. Although it is considered “late-onset”, the signs of deterioration can be seen as early as five months of age in some cases. To understand how this disease is inherited, we must first remember that females have two X chromosomes (XX), while males have one X and one Y chromosome (XY). In XL-PRA1, the defective gene for PRA is a recessive trait which is found on the X chromosome. Females who inherit a defective gene on the X chromosome from one parent and a normal gene on the X from the other parent will be carriers with partial retinal defects. These defects are so subtle that the dog’s vision will not be affected. They do however have the ability to silently pass the defective gene on to their offspring. The males are different due to their XY make up. A male puppy with a mother who is a carrier is either going to inherit a defective gene or a normal gene depending on which copy of the X chromosome they inherit from their mother. If it is a defective gene, the dog will be affected because in X-Linked diseases, there is no such thing as a male carrier. For this reason, male sibes are much more likely to be affected than females. As female carriers (“heterozygous dogs”) have a 50% chance of passing a defective gene on to their offspring and any male offspring who inherit the defective gene will be affected by the disease, only clear females should be bred from. Clear females (“homozygous normals”) can only pass a normal gene on to their offspring. Thankfully Optigen has developed a genetic test to accurately detect the genetic status of each dog enabling breeders to make responsible choices in their breeding programs. There are 3 different genetic statuses for female dogs; normal (XnXn), carrier (XnXa) and affected (XaXa) but only two genetic results for males; normal (XnT) and affected (XaY). Below are the statistics for different breeding combinations. While these are the statistics, not all litters will adhere to the numbers and may produce results that differ from the numbers shown. PRCD-PRA (Progressive Rod Cone Degeneration Progressive Retinal Atrophy)
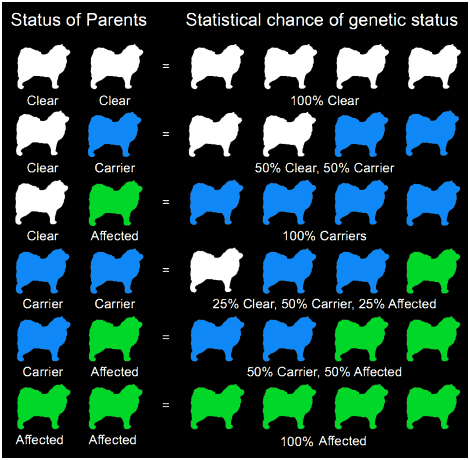
Though it is not the most common eye disease in Lapphunds (affecting less than 2%, with fewer and fewer diagnoses since the development of a genetic test), PRCD-PRA is one of the most well known as most breeders will test at least one parent, if not both parents for their genetic status and will often advertise this status in litter announcements and on individual dogs’ pages on their websites. Though the disease causes the same symptoms as XL-PRA1, the mode of inheritance is different as it does not depend on the X chromosome and males and females are statistically equal in terms of inheritance. The age of on-set is generally slightly later too, with most Lapphunds not showing clinical signs of the disease until they are 5-8 years old. Officially, the mode of inheritance of PRCD-PRA is known as autosomal recessive, meaning that both parents have to carry at least one copy of the mutant (affected) gene in order for their offspring to be genetically affected. Thankfully a test has also been developed for PRCD-PRA so breeders can make informed decisions when it comes to their breeding programs. Genetically dogs can have one of three statuses: Normal – dog carries two normal copies of the gene and cannot pass an affected gene on to their offspring. Genetically normal dogs will not develop PRCD-PRA. Carrier – dog carries one normal and one mutant (affected) copy of the gene and has a 50% chance of passing on a mutant gene to their offspring. Genetic carriers will not develop PRCD-PRA. Affected – the dog carries two mutant (affected) copies of the gene and can only pass an affected gene on to their offspring. Genetically affected dogs may develop PRCD-PRA. Below are the statistics for different breeding combinations. While these are the statistics, not all litters will adhere to the rules and may produce results that differ from the numbers shown. As the gene pool for Lapphunds is relatively limited, electing to remove carriers of PRCD-PRA from breeding programs (particularly in our much smaller gene pool in Australia) would result in the loss of very valuable genetic diversity, though some breeders in Australia have chosen to do so. While most breeders test very early in Australia and will test an entire litter for their genetic status, it is uncommon for these tests to be conducted until later on in Finland, with breeders preferring to see how their potential breeding stock will turn out before having them tested. It is not uncommon for breeders internationally to only test breeding stock rather than the entire litter.
|
Glycogen storage disease type II (Pompes)
Glycogen Storage Disease Type II, or “Pompes” Disease is a condition where
there is a build up of a form of glucose, called glycogen, in the body. In most
dogs, there is an enzyme responsible for metabolising glycogen, but in dogs
with Pompes, this enzyme is absent, causing glycogen to accumulate in the muscle
and nerve endings, impeding their normal functions and causing irreparable damage.
The symptoms of Pompes vary a great deal from dog to dog and unfortunately there is no cure. Often dogs will show signs of progressive muscle weakness, frequent vomiting and regurgitation (due to a dilated esophagus), nonstop panting and heart abnormalities. Dogs affected by Pompes are usually diagnosed at around 6 months of age and often don't live past their first birthday. Sadly, this disease has been found in the Lappish breeds, though a genetic test for the disease has been developed, meaning that dogs can now be tested for their genetic status, either clear (where the dog has two normal copies of the gene – this dog cannot develop the disease but if bred to a carrier, may have puppies who are carriers of the disease), carrier (one normal and one mutated copy – this dog will not develop pompes disease but if bred to a dog who is also a carrier, or who is affected their puppies may develop the disease) or affected (two mutated copies of the gene – this dog is likely to develop pompes disease). Like PRCD-PRA and DM, Pompes is autosomal recessive (see diagram above) meaning: Clear/Clear -100% of offspring will be clear Clear/Carrier – Each puppy has a 50% chance of being clear, and a 50% chance of being a carrier. Clear/Affected – 100% of offspring will be carriers Carrier/Affected – Each puppy has a 50% chance of being a carrier, and a 50% chance of being affected. Affected/Affected – 100% of offspring will be affected. |
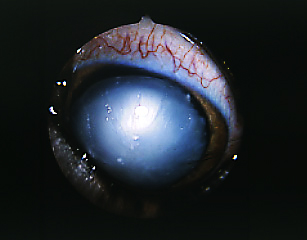
Hereditary Cataracts
There are many, many different forms of cataracts that can affect dogs, each with a different cause, age of onset and speed of progression. In Lapphunds and Sibes, hereditary cataracts usually appear after the age of one year, however, they may not appear until the dog is at an advanced age.
As with many diseases, unfortunately the genetic inheritance of hereditary cataracts in Lapphunds and Sibes has not yet been found, though it is believed to be autosomal recessive (the same as PRCD-PRA and DM). For this reason, any dog who is to be bred from, should have regular eye checks (ask to see the parents of your potential puppy's ACES test documents). There is research underway to identify the faulty genes that cause hereditary cataracts and it is hoped that in the future, we will be able to screen for the disease through genetic testing. Cataracts are a clouding of the eye's lens, caused by a breakdown of tissue within the eye. The lens focuses light onto the retina which allows the dog to see. As cataracts cause the lens to become cloudy, it diffuses the light, limiting the dog's ability to see. It generally begins limited or fuzzy vision and often progresses to total blindness if not treated. Surgery is an option, however it is often highly costly and sometimes, not successful. Cataracts can affect one or both eyes. |
Canine Degenerative Mylopathy (DM)
Canine Degenerative Myelopathy or DM is an adult-onset disease of the spinal cord that affects both purebred and mixed breed dogs. DM causes degeneration of the “white matter” of the spinal cord and the nerves around it. This white matter contains fibres that transmit movement commands from the brain to the rear extremities as well as sensory information from the limbs to the brain.
This loss of sensory information means that while it is not a painful disease in itself, over time, it causes loss of sensation, loss of limb control and in the advanced stages, paralysis. Because of the impairment of the dogs’ hind legs, compensatory movements can cause pain in the dog’s front end from stress, overuse and/or abnormal repetitive movements. Typical onset is between 8-14 years of age and both dogs and bitches are equally affected. Unfortunately there is no cure for DM, though physical therapies and certain medications can increase the dogs’ life expectancy from 6 months – 1 year up to two - three years. The disease is a genetic disorder, and a mutation of the SOD1 gene has been identified as a major risk factor for the development of DM. A genetic test was developed in August 2009 to identify those dogs who are at major risk of developing the disease and several laboratories suggest the test for dogs of all breeds, as the faulty gene appears to have originated in ancient dogs before they were domesticated. As such, it is now recognised as being in over 50 breeds. Unfortunately many cases of DM go undiagnosed because there is no test to specifically identify the disease. The diagnosis of DM is a process of elimination, meaning that your Veterinarian will look at other, more treatable and more easily identified diseases, injures and conditions before they will look at DM. The only way it can be conclusively diagnosed is on necropsy (an autopsy performed after the dog has passed away), when a small amount of the spinal cord can be removed and examined under a microscope to check for the classic histopathologic lesions of DM. As many owners do not wish to have the necropsy performed on their beloved pet, there are many deaths caused by a condition that is suspected to be DM, but not diagnosed as such. Given the age of onset, there are many dogs who have not lived through the diagnostic process or whose symptoms have been attributed to old age, rather than a specific disease. There are many diagnosed cases of DM in Siberian Huskies, though it is seen less and less with the advent of DNA testing and the understanding of it's mode of inheritance, which is identical to that of PRA. A video of a Siberian Husky tested to have a genetic status of "affected" (meaning they carry two faulty copies of the SOD1 gene) can be seen here (fast forward to roughly 1:20 into the video to watch the movement of the dog), though due to the disease not being able to be conclusively diagnosed until after death, it is only suspected that the dog has DM at this point. While there have been no documented diagnoses of the disease in Finnish Lapphunds, the faulty SOD1 gene that has been found to put dogs of other breeds at high risk of developing DM, has also been found in Finnish Lapphunds. Currently, there have been no Finnish Lapphunds who have tested as affected, though the test is not often carried out on Lapphunds. With testing, it is hoped that Lappie breeders will have the information they need to suitably manage the risk of breeding affected dogs. In order to develop the test for all breeds, over 6600 canine DNA samples studied, 31 of which were Finnish Lapphunds. Of these 31 Finnish Lapphunds, 27 of them tested as Normal/Clear (87.1%) and 4 tested as Carriers (12.9%). Because the faulty gene was present amongst 57 of the 147 breeds tested, it is believed that the gene is not breed specific, and therefore, a breed specific marker for the disease does not need to be developed. Because there has not been a diagnosed case of DM in Finnish Lapphunds, it is impossible to know what would happen to an affected dog. It is possible that other genetic attributes, environmental conditions etc could inhibit the disease from ever manifesting in Lapphunds. It is also possible however that an affected Lapphund would develop the disease in the same way other breeds do. For this reason, it is recommended that breeders of Finnish Lapphunds manage the risk of breeding affected dogs by testing their breeding stock. |
|
NSW Breeder Identification Number: B000685661 | Victoria Source Number: EE102260
Dogs NSW Member Number: 2100088743 |
© Pawformance 2023
|